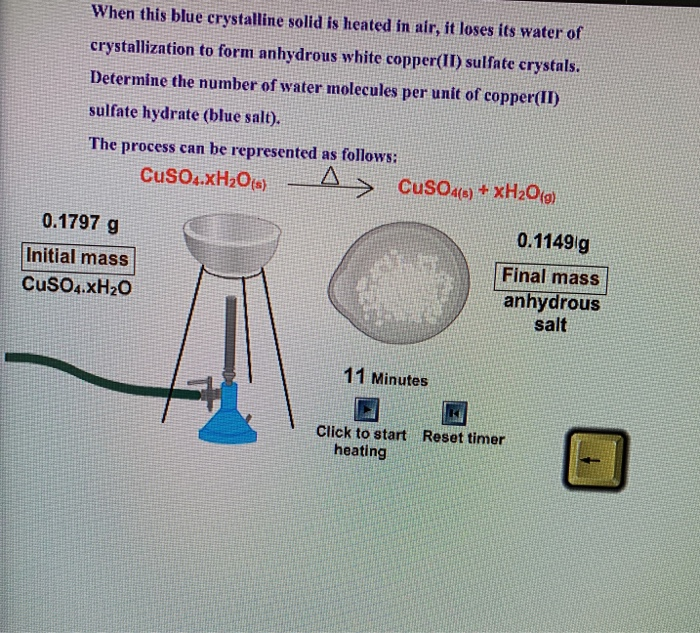
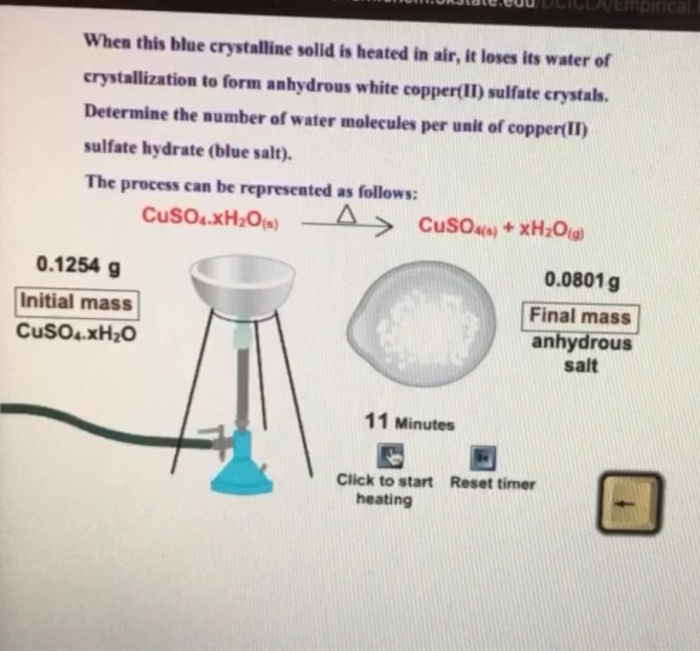
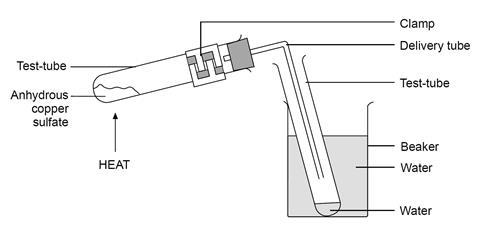
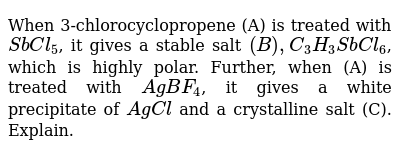If the crystalline salt NA2SO4.xH2O on heating loses 55.9% of its mass, then what is the formula of the salt? - Quora

The metal salt A is in blue colour. When salt A is heated strongly over a burner then a substance B present in it is eliminated and a white powder C is

A white crystalline solid P on strong heating decomposes to give a reddish brown gas R and residue Q - Brainly.in
NaCl-related weathering of stone: the importance of kinetics and salt mixtures in environmental risk assessment | Heritage Science | Full Text

Molecules | Free Full-Text | Molecular Crystallization Inhibitors for Salt Damage Control in Porous Materials: An Overview | HTML

A crystalline salt when heated becomes anhydrous and losses 51.2% - CBSE Class 11 Chemistry - Learn CBSE Forum

What is water of crystallisation Explain that crystalline salts contain water of crystallisation - Chemistry - Acids Bases and Salts - 16253635 | Meritnation.com

the crystallisation salt na2so4xh2o on heating losses 559 of its weight the formula of the crystalline salt is - Chemistry - TopperLearning.com | cf614k66